Intrinsically disordered regions (IDRs) are ubiquitous stretches of protein that do not adopt a stable structure, are a major class of protein structure found in all living organisms and viruses, and are predicted to be present in around a third of eukaryotic proteins.
IDRs can, in a regulated manner: (i) undergo disorder-order transition to adopt a functional, structured conformation, e.g. through protein-protein interactions (PPIs) involving one or two IDR partners; (ii) contribute to phase separation promoting assembly of membraneless organelles and signalling complexes; or, (iii) regulate the function of adjacent ordered domains of the protein in which they lie. The timeframe upon which dynamic conformational transitions occur affects a protein’s interactome and, therefore, crucially, its function. IDRs and their order-disorder transitions regulate diverse functional proteins (and thus cellular processes), incl. transcription factor (TF) complexes, chaperones, DNA damage repair complexes, cytoskeleton scaffolding, and import/export complexes. IDRs are important and overrepresented in key signalling and disease pathways. For all these classes of protein, understanding and targeting IDR-dependent PPI interactomes with selective and spatiotemporal resolution would provide a major step forward in our understanding of biological mechanisms and, in the longer term, our ability to develop targeted interventions.
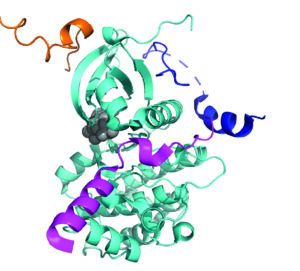
This research programme focuses on a specific system of IDRs that regulate a range of vital cellular functions through a common factor, the protein kinase Aurora-A. Each IDR in this system achieves a distinct function by localizing Aurora-A to a different subcellular compartment, regulating its catalytic activity, and forming a multivalent complex that brings additional components of the relevant pathway into kinase proximity. Our goals are to ascertain how different IDR partners bind Aurora-A’s surface, how the structure and dynamics of IDRs alter upon Aurora-A interaction, and how the properties of the IDRs and their interactions relate to the dynamics of Aurora-A localisation and activation in cells. Contextualizing molecular mechanisms within the framework of Aurora-A’s interactome to understand functional outcome, will allow us to:
- Assign IDR-dependent PPIs of Aurora-A to a functional outcome, leading to new methods for mechanistically precise interventions to disrupt them.
- Design and generate IDR-specific chemical and biological tools to dissect complex cellular mechanisms.
- Deliver broader understanding of how the dynamics of IDRs direct and scaffold PPIs in distinct locations to achieve spatially and temporally specific activities.
